The New South Wales gold rush began more than 400 million years ago.
It was an age of fire, that ended with ice. Australia was part of the super-continent Gondwana, which was not yet south. By continent standards it was moving fast. By the end of this era, the Ordovician, it would be at the south pole.
In the water there were snails, trilobites, corals and some ‘primitive’ fish. On the land, some little bug type fellas with exoskeletons were starting to colonise Gondwana, which was otherwise empty of critters.

Volcanoes ruled a section of Gondwana that mining companies now refer lustfully (or covetously) as the LFB, the Lachlan Fold Belt. Deep in the earth, gold was already there. Hot magma rose upwards towards the surface, releasing fluids as it went. Amidst these fluids was liquid gold. Gold poured onto the surface of Gondwana, producing substantial reefs of golden metal due to volcanic ‘intrusions’ and also thin streaks through the granite. Then there was the Devonian , which was the Age of the Fishes, because fish came to be on top of the earth’s food chain (if one can have a favourite geological era this is mine). In this era, some of this gold was disrupted by more geological activity, so that gold was enfolded in quartz. This was the gold that diggers began to extract by the rivers in Ophir and Hill End in 1851.
What are we to make of this gold? Perhaps it was waiting patiently under the earth for its purpose to be fulfilled, when finally, the combinations of the end of the Napoleonic Wars, new British banking legislation, imperial expansion, steam ships and settler colonial confidence let thousands of people to take the three-week trip from Sydney to Bathurst and the eight hour journey to extract the gold.
Which is to say, was it always gold, not in terms of composition ot substance, but potential? Was the ‘gold’ sought by diggers and the bankers who bought it from them, embodied in what the magma deposited? Or did the diggers bring gold – the idea of gold – with them? And was it that idea which was infused with the historical moment, the confluence of a specie shortage, the disciplining of bank money and increased global mobility – so much for those diggers to carry! – that made gold, gold?
This is clearly important. Check out the price of gold over one hundred years (source: https://www.macrotrends.net/).
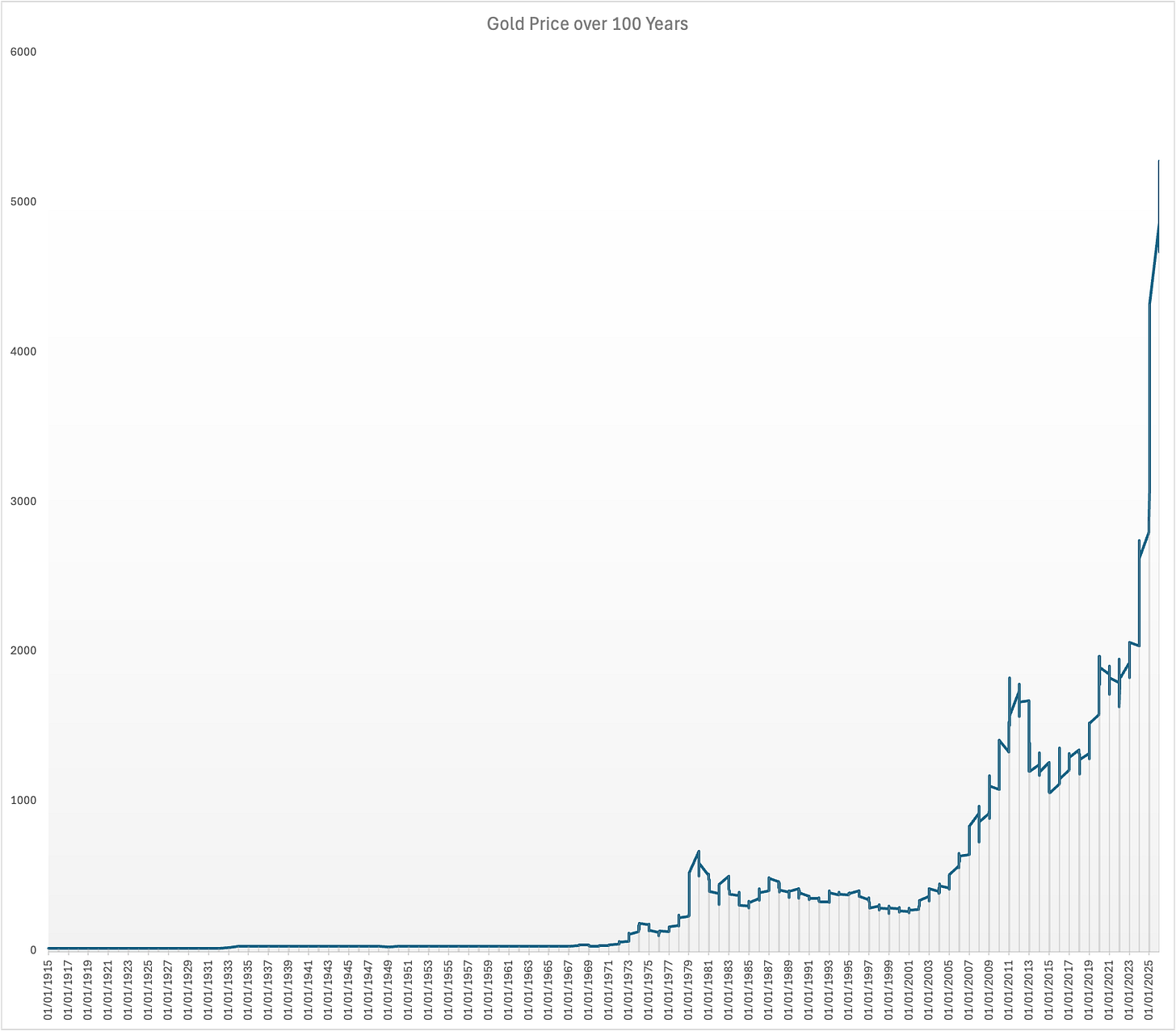
There was the gold standard and dollar-gold convertibility until 1969. Then, the moment that gold wasn’t important (in one way of thinking about it), the price goes nuts. And it goes nuts, as we have just seen, because of lots of ideas about gold: a safe haven when other currencies look dodgy, an opportunity for short-term gains in the process etc.
The relationship between the idea and the price is obvious.
But it is not just the idea of gold, is it? The point of it being gold is that it is actually, materially gold. Of course other metals are also important, but even in bimetallic systems of currency, while you can exchange their value like we do types of currency, you can’t actually substitute silver or cooper or anything else for gold. It actually has to be gold.
No wonder we have so many stories of people able to turn something that isn’t gold, into gold. This was the ancient science/philosophy (when those two were pretty much the same thing) of alchemy – or, at least, a sub-branch of alchemy called chrysopoeia, which distinguishes the gold-making branch from spiritual uplift/elixir of life type branches (though some scholars point out that these were in fact entangled).
It was also the gift/curse of Midas in Greek mythology, whose touch turned everything to gold. And then there is Rumpelstiltskin, who could spin straw into gold.
Of course, all such things are mythical. Gold cannot be magicked into being. There are a few cases of sciencing it into being, but in at least one case it was radioactive. That would probably impact its price, I reckon.
What about 400 million years ago? Did the Ordovician volcano perform primodial alchemy?
Turns out, no. The gold was already there.
Recently, astrophysicists have been able to prove that gold is created when stars collide.
I mean this seems like fucking nonsense, doesn’t it? But seriously. Here is how you make gold.
First you need not just one dead star, but two. Getting hold of two dead stars is quite difficult, but I reckon with some perseverance we can make it work.
Specifically they have to be ‘neutron stars’, which sounds like a star wearing a Superman cape, but is just the ultra-dense remnants left over when a star dies.
Then you need the two stars to collide. Not just line up in the sky when you’re looking at them – we’re making GOLD here, not the zodiac.
It is tricky to do, but the payoff is great. One 2017 collision produced 3 and 13 earth-masses of gold (that’s a pretty wide margin of error, but obv the low end is fine).
How do we get it to earth though? Well this is the really tricky bit. The gold that the Ordovician volcano reached down and extracted from the earth’s core and deposited on the LFB. Well, it was in the dust that coalesced to form the earth itself. When the earth was made of molten stuff, the gold sank deep into the core. Bits of gold dust have sometimes fallen from space, but most of it was produced by the cosmic alchemy and then helped form the earth itself.
Suddenly, today’s price ($4,650.80 USD an ounce) seems a bit of a bargain.
This post is part of my CH Currey Memorial Fellowship at the State Library of NSW on ‘What happened to the gold’?
{ 6 comments… read them below or add one }
Ken_L 04.10.26 at 6:44 am
Once Elon figures out how to kill stars stone dead and install collision apps in them, his Mars colony will have more gold than the Oval Office.
oldster 04.10.26 at 9:41 am
Fun piece! Thanks.
Sean 04.10.26 at 5:17 pm
I think that chart is in nominal dollars. When a US dollar is defined as equal to a certain weight of gold, its a tautology that the price of gold in USD is stable. The same site shows inflation-adjusted prices moving dramatically in the era of the gold standard.
There are archaeologists who talk about the indigenous peoples north of Mexico having no interest in gold even when they had access. It just did not speak to them the way it did to the Mexica or the Scythians, although some of them were happy to mine or pan when they met people who would buy it.
J-D 04.11.26 at 8:20 am
For some reason, Youtube has served me a lot of ads for gold. I haven’t seen how any of them end because I’ve always been able to skip, but after a while this question has formed in my mind: ‘If you think this stuff is so great, why are you trying to sell it to me, instead of hanging on to it?’
Doug M. 04.11.26 at 11:01 am
Oooh oooh ooh, this touches like three of my favorite nerd-obsessions. Thank you, Hannah.
Let’s see. Number one is, the discovery that gold and a bunch of other heavy elements were (mostly) formed by colliding neutron stars? That’s relatively recent, and it pretty much follows the classic pattern of a Kuhn Paradigm Shift:
— there’s a theory that explains stuff (heavy elements beyond iron are formed in supernova explosions)
— however, there’s a weak spot in the theory (it’s unclear how a supernova explosion can add enough neutrons, fast enough, to create certain elements and isotopes)
— over time, the weak spot becomes a hole (we learn more about supernovae, we understand nuclear physics better, there’s no way supernovae can create the cosmic abundances of elements that we see in the universe. they’re just not producing neutrons fast enough)
— a bunch of theories and ideas are propounded. One of them is fringe, bordering on ridiculous (you know what would produce a lot of neutrons very fast? colliding neutron stars!)
— the theory does not win broad acceptance (ha ha, colliding neutron stars. what are the odds?) but clings to life
— gradually, evidence accumulates to favor the theory (actually, we’re seeing events that might be colliding neutron stars. wait, LIGO is detecting events that are /definitely/ colliding neutron stars. wait, it looks like colliding neutron stars are pretty common, actually.*)
— a smoking gun is found (we’ve observed a neutron star collision throwing several Earth masses of gold into the interstellar medium)
— over a generation, theory moves from fringe to serious to accepted consensus / fact.
So that’s pretty neat!
*Colliding neutron stars are more common than we initially thought because higher-mass stars are more likely to be binaries or otherwise part of multiple-star systems. So, if a star is big enough to become a neutron star, it’s more likely that it has a companion that’s also big. And if that’s the case, and they’re close enough, they’ll gradually spiral inwards towards each other through some combination of interacting accretion disks and radiation of gravitational waves.
It’s still not exactly a /common/ set of circumstances. But in a big old universe? it’s going to happen often enough.
Cool post, thank you! will come back to nerd out a little more if time allows.
Doug M.
Doug Muir 04.11.26 at 3:49 pm
Nerd comment #2: The Goldschmidt Classification.
Wikipedia actually has a solid article on this, if you’re interested. But here’s the TLDR: there’s a big difference between the abundance of elements in the Solar System, and the abundances of elements here on Earth. And then there’s a /different/ set of differences between the abundances in the Earth’s crust vs. the Earth’s mantle and core. And the reason for this is that most elements fall into a few groups — the “Goldschmidt Classification” — determined by how they behaved during the Earth’s formation.
So for instance: most metals form oxides. And most metal oxides have this interesting property: they’re lighter — less dense — than the original metal. So, for instance, rust is about 2/3 as dense as iron.
(This is why rebar must be pretreated against corrosion. You build a rebar frame and pour concrete around it to build a bridge or whatever. But then there’s a little crack and a tiny bit of water gets in and reaches the rebar. Once the rebar starts to rust, it will expand, because rust is less dense than iron or steel. And this will crack the concrete further, allowing in more water, in a vicious cycle that can end with your bridge falling down.)
The Earth’s crust — I am oversimplifying here, but this is basically correct — is made of the stuff that was lighter, and that floated to the top during Earth’s formation and initial cooling. And because oxides tend to be lighter than their parent metals, this light stuff included a lot of oxides, particularly of metals and metalloids. Pick up a random rock: it’s almost certainly some combination of oxides, most likely of silicon, magnesium, aluminum, calcium, or iron.
There are a bunch of interesting twists and exceptions, because chemistry. So, there are some elements where the dominant form in the crust is some different compound that’s not an oxide — like, lead ore is a light sulfide rather than a light oxide, and calcium is likely to hang around as a carbonate (limestone). And there are elements that form light oxides but that were lost from the formation of Earth for other reasons. But “form a lighter compound, usually an oxide, and that compound is more likely to stay in the Earth’s crust” is the broad general pattern.
Okay so the thing about gold and the other “noble metals” — platinum, iridium, those guys — is that they don’t form oxides at all. And most of them don’t form other compounds, chlorides or sulfides or whatever, very enthusiastically either. So they mostly stay in their pure elemental form. Which is nice, but it means that — since they’re dense metals, a lot denser than the rock of the crust — there’s nothing to stop them from sinking downwards into the mantle and core.
So gold and the other noble metals should be a lot more common elsewhere in the Solar System than on the surface of the Earth. And in fact this seems to be the case. You may have heard of the famous “iridium layer” that marks the extinction of the dinosaurs? A lot of asteroids — not all, but a lot — appear to be relatively enriched in noble metals.
(Relatively enriched. Like, if an asteroid has 100X more gold than the Earth’s crust, you’re still not going to stumble over gold nuggets on its surface. 100X very very little is still not much.)
Fortunately for us, there are geological processes — volcanism, water, weathering — that can concentrate the small amounts of gold in the crust. A planet without those processes would probably have a lot fewer useful ore deposits… but that probably deserves a blog post of its own some time.
Doug M.